By Priyanka Sharma
With the COVID-19 vaccination just around the corner, the Union Health Ministry on Thursday said that vaccination for COVID-19 will be voluntary.
The Health Ministry said that it is advisable to receive the complete schedule of COVID-19 vaccine for protecting oneself against this disease and also to limit the spread of this disease to close contacts including family members, friends, relatives and co-workers.
The Ministry further informed that two doses of vaccine, 28 days apart, need to be taken by an individual to complete the vaccination schedule.
For the mass vaccination as per the potential availability of vaccines, the Centre has selected the priority groups who will be vaccinated on priority as they are at higher risk.
“The first group includes healthcare and frontline workers. The second group to receive COVID 19 vaccine will be persons over 50 years of age and persons under 50 years with comorbid conditions,” stated a health ministry document on FAQ related to the COVID-19 vaccine.
Responding to a question whether an infected person could get the vaccine, the health ministry noted that infected individuals should defer vaccination for 14 days after symptom resolution.
The Centre said that the eligible beneficiaries will be informed through their registered mobile number regarding the health facility where the vaccination will be provided and the scheduled time for it.
The registration of the beneficiary is mandatory for vaccination for COVID-19. “For the verification document for vaccination, a photo ID is a must for both registration and verification of beneficiary at session site to ensure that the intended person is vaccinated,” noted the government document.
Recently ANI had reported that Union Health Secretary Rajesh Bhushan informed there were nine COVID-19 vaccine candidates in different phases of development in India, of which three are in pre-clinical phase whereas six are under clinical trials.
“Some of them may get licensed in the coming weeks but we cannot foretell at this moment of time because licensing is the domain of the national regulator DCGI,” stated Bhushan adding that people still have to maintain Covid precautionary measures even when the vaccines will be available as recommended by scientists around the world.
The first vaccine is Covishield being manufactured on the Chimpanzee Adenovirus platform and is being manufactured by the Serum Institute of India (SII) in Pune. The collaborative company is Astrazeneca. The regulator had allowed the phase 2 and 3 clubbed clinical trials of the vaccine to them and they have applied for emergency use authorisation,” he said.
The second vaccine is Covaxin which is on an inactivated virus platform. This is being developed by Bharat Biotech International Ltd, Hyderabad. Their collaborator is the Indian Council of Medical Research (ICMR). This is in phase 3 trials. They have also applied for emergency use authorisation.”
The third vaccine candidate is ZyCoV-D which is based on the DNA platform. It is being manufactured by Cadila Healthcare Ltd, Ahmedabad. Their collaborator is the Department of Biotechnology. It is in phase 3 of trials, he informed.
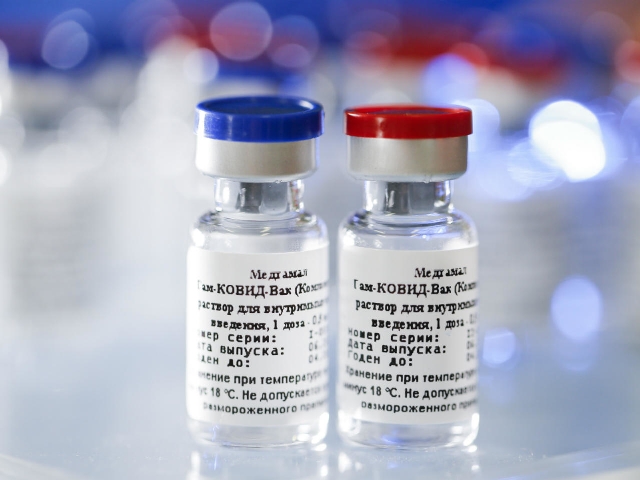
The fourth vaccine candidate is the Russian vaccine (Sputnik V) which is based on the human adenovirus platform. It is being manufactured by Dr Reddy’s Lab in Hyderabad. Their collaborator is Gamaleya National Centre, Russia and the vaccine is in the phase 3 trial.
The fifth vaccine candidate is NVX-CoV2373 which is based on the protein sub-unit platform. It is being developed by the Serum Institute of India, Pune. Their collaborator is Novavax. The phase 3 trial of this vaccine is under consideration in India.
The sixth vaccine candidate is a recombinant protein antigen-based vaccine. It is being manufactured by Biological E Ltd, Hyderabad. Their collaborator is MIT, USA. Its phase I and II human clinical trials have started. The seventh vaccine candidate is HGCO 19 which is an mRNA-based vaccine. It is being manufactured by Genova, Pune. Their collaborator is HDT, USA. The pre-clinical animal studies are over for this vaccine. Phase 1 and 2 clinical trials will start for this vaccine in the near future.
The eighth vaccine candidate is an inactivated rabies vector platform. It is being manufactured by Bharat Biotech International Ltd, Hyderabad. Their collaborator is Thomas Jefferson University, USA. It is in the advanced pre-clinical trial stage.
Vesiculo Vax platform manufactured by Aurobindo Pharma in collaboration with Aurovaccine (USA) is in the pre-development stage. (ANI)